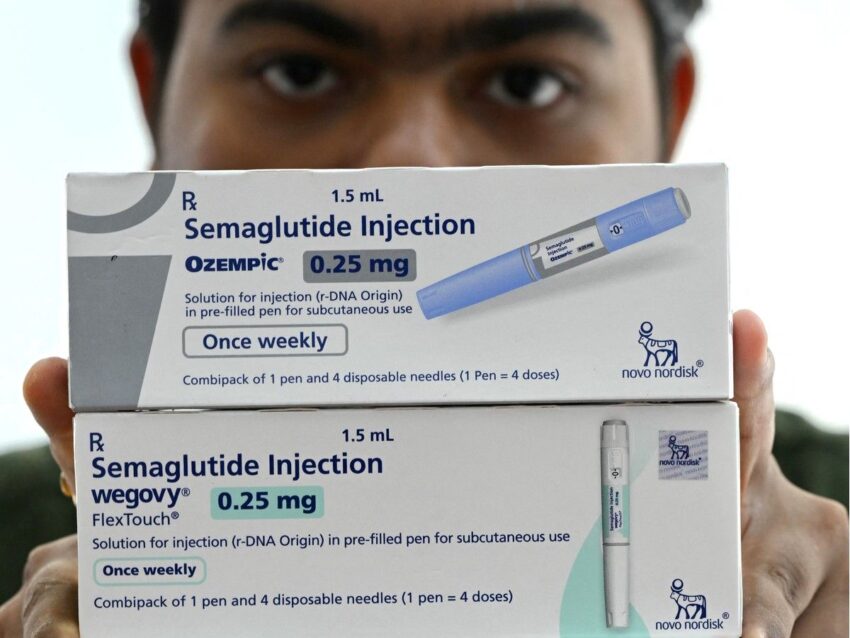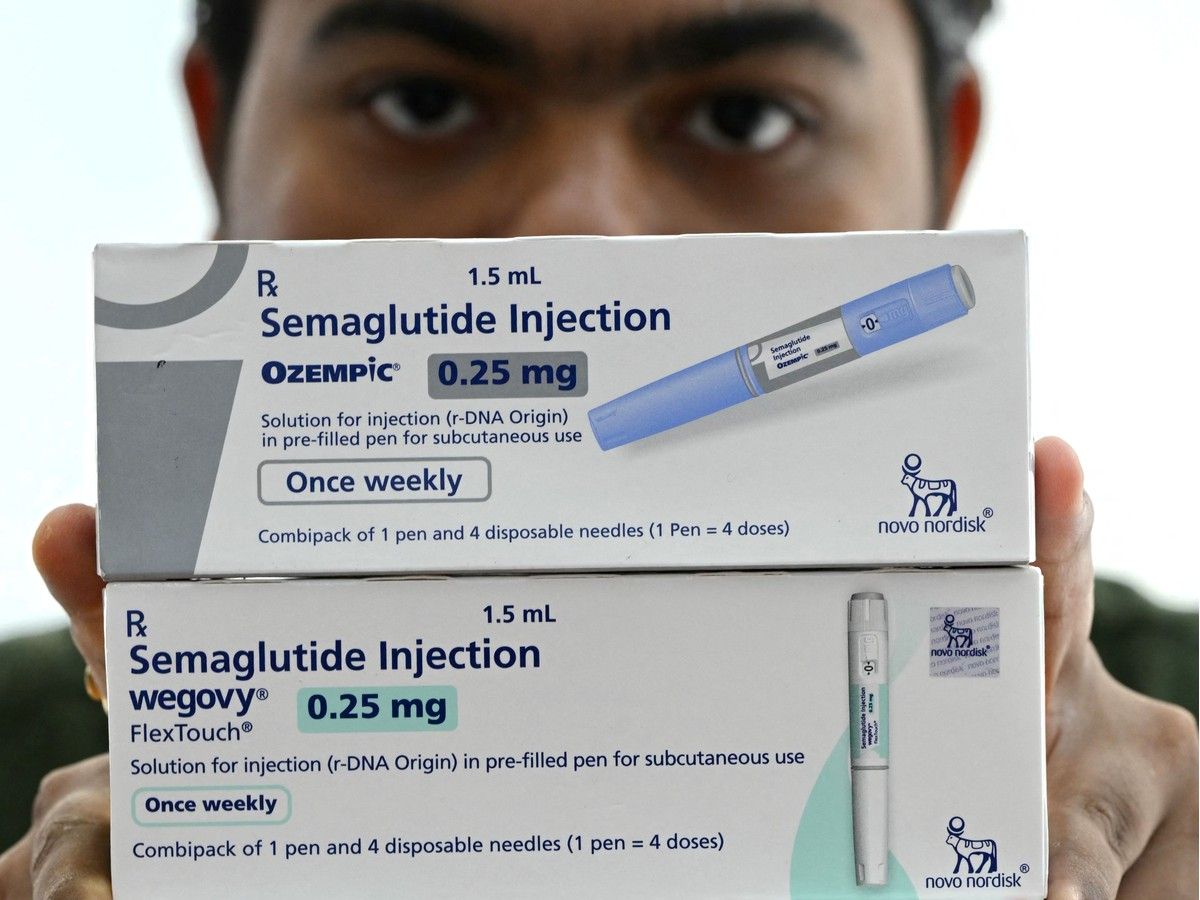
With the
expiration of the Canadian patents
for weight-loss drugs Ozempic and Wegovy in January, experts are looking to mid-summer for cheaper generic replacements to come on the market.
The patents kept the cost of those drugs high because Danish drug manufacturer
was the only company that could make them.
Patients in Canada have been paying between $300 and $400 a month, depending on the prescribed dose.
Dr. Sanjeev Sockalingam, scientific director of Obesity Canada, says
accessibility to these medications is a significant consideration
for his patients. The cost “limits one tool in our toolbox for obesity management.”
These drugs mimic a natural gut hormone to help control blood sugar levels, slow digestion and increase feelings of fullness.
says it is reviewing nine submissions for generic versions of the popular weight loss medications. The submissions are for generic forms of semaglutide, the active ingredient in Novo Nordisk’s injectable drugs Ozempic and Wegovy, the former approved by Health Canada for diabetes and the latter for weight loss.
The government department doesn’t give a timeline regarding when generic forms of these drugs will be approved, but Mina Tadrous, associate professor of pharmacy at the University of Toronto, told CBC News that
generics might be available by this summer or early fall
.
Meanwhile, in India, several companies obtained early approval for generic replacements and began selling them the day the patents expired. One Indian company was offering generics for as little as C$19 a month. Analysts expect around 50 generics could enter the Indian market within months.
Tadrous says India may be a major supplier of pharmaceuticals to Canada, but the generics still have to go through Health Canada’s approval process before consumers can buy them.
Swiss generic drugmaker Sandoz told
last November that unbranded versions of Ozempic should launch in Canada by the end of June.
The company is hoping to be one of the first entrants in the Canadian market, after the expiration of Novo Nordisk’s patent for semaglutide expires. The regulatory process is underway for Sandoz, as well as Apotex, Teva Canada, Taro Pharmaceuticals and Aspen Pharmacare Canada.
Our website is the place for the latest breaking news, exclusive scoops, longreads and provocative commentary. Please bookmark nationalpost.com and sign up for our daily newsletter, Posted, here.